Kat KelleyGHTC
Kat Kelly is a senior program assistant at GHTC who supports GHTC's communications and member engagement activities.
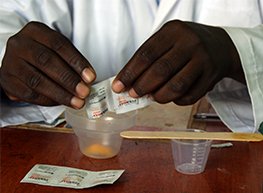
After four successful Phase 3 clinical trials enrolling more than 3,500 patients in 18 countries, the European Medicines Agency (EMA) has granted Pyramax® Granules, a pediatric malaria medicine, a positive opinion under Article 58—a mechanism through which the EMA reviews and issues a scientific opinion on products that are not intended for use in Europe. Pyramax® Granules is a combination of two antimalarials and was developed by Medicines for Malaria Venture (MMV), a member of the GHTC, in partnership with Shin Poong Pharmaceutical Co. Ltd. The treatment is designed to be easy for children to consume: its bitter taste is masked and it is taken in liquid form, as children often struggle to swallow pills. Next, MMV and Shin Poong will work to introduce Pyramax® Granules as well as Pyramax®, the tablet version which is suitable for adults, in malaria-endemic countries.
A recent article in NPR: Goats & Soda examines Zika virus, a disease transmitted by the same mosquito vector of dengue fever and chikungunya, that has been rapidly spreading throughout South America and the Pacific Islands in recent years. One in five individuals infected with the virus experience Zika fever, which in addition to fever involves rashes, joint pain, conjunctivitis, headaches, vomiting, and muscle pain. Cases of Zika fever are likely underdiagnosed, as awareness of the disease is limited and its symptoms are similar to infections associated with other members of the Flaviviridae virus family, including dengue fever, yellow fever, West Nile, and Japanese encephalitis. There are no reported deaths to date, but there is growing evidence that the virus can be transmitted sexually or from mother to infant during pregnancy. As there are no vaccines or drugs for Zika fever, treatment involves addressing the symptoms, using fever reducers and painkillers.
A group of global health advocacy organizations—including GHTC members the Drugs for Neglected Diseases initiative, TB Alliance, Infectious Diseases Society of America Center for Global Health Policy, Sabin Vaccine Institute, and Treatment Action Group—sent an open letter to the US Senate Health, Education, Labor and Pensions (HELP) committee, calling for amendments to the priority review voucher (PRV) program at the US Food and Drug Administration (FDA) to ensure it adequately incentivizes research and development (R&D) for neglected diseases, as intended. Under the program, companies that receive FDA approval for a drug for a neglected disease (see list of eligible diseases) are awarded a voucher for expedited FDA review (six months rather than ten) of a future product. The vouchers are transferable and have been sold at increasing rates, the most recent going for US$350 million. The letter calls for PRVs to only be awarded for products that are "truly new" as two of the six vouchers that have been awarded were for products that had long been in use outside of the United States.[1] The organizations are also calling for recipients of the voucher to be required to develop a strategy to ensure the product is made affordable and accessible to patients in the countries in which the eligible disease is endemic.
The United Kingdom (UK) announced last week the creation of the Ross Fund which will provide £1 billion ($1.5 billion) over five years to fight malaria and other infectious diseases. The funding, which will come from the UK overseas aid budget, includes £90 million for malaria eradication; £100 million for infectious disease R&D; £115 million for the development of drugs, diagnostic, and insecticides for malaria, tuberculosis, and other infectious disease resistance; and £188 million for biodefense and rapid response systems for epidemic preparedness.
Global Good has developed a cooler that can store and transport vaccines at freezing temperatures for over a month using just ice and no electricity. The Arktek cooler uses vacuum thermal insulation, an approach used to preserve spacecraft entering extreme temperatures. The cooler has been tested in Senegal (with support from GHTC member PATH) and Ethiopia, and last year, the Arktek was modified for use in Ebola vaccine clinical trials, during which the cooler kept the vaccines between -76 and -112 degrees Fahrenheit for three days.
[1] There are two separate PRV programs at the FDA, one for neglected tropical diseases and one for rare pediatric diseases. Of the six PRVs that have been awarded, three each have been for neglected tropical diseases and rare pediatric diseases. Since the beginning of the PRV program, six PRVs have been awarded—three for neglected tropical diseases and three for rare pediatric diseases.