Claire WingfieldPATH
Claire Wingfield is a Product Development Policy Officer within the Advocacy and Public Policy Department at PATH, a global nonprofit dedicated to ending health inequity.
In this guest post, Claire Wingfield—product development policy officer at PATH—writes about a new paper that examines partnerships between the public and private sectors to advance global health innovation, research, and development.
For more than 30 years, PATH has been working to advance innovative global health solutions. Partnering is a core part of PATH’s strategy and is fundamental to how we develop and deliver high-impact, cost-effective technologies. Our partners come from international health agencies, governments, universities, other nongovernmental organizations (NGOs), and private-sector companies. Each partnership is unique, but no matter who the partner is, the collaboration must be mutually beneficial and fulfill our mission to improve the health of people around the world.
Public-private partnerships provide a forum for maximizing complementary areas of expertise. They improve knowledge of the most pressing global health needs and lead to solutions that improve health equity. By strengthening the capacity of different sectors, these partnerships accelerate the development and introduction of the most promising technologies for those most in need. Finally, forging new cross-sector partnerships with public institutions and commercial entities enable organizations like PATH to maximize the limited global health resources of both sectors—financial, technical, and structural—to reduce costs and avoid redundancy in our efforts to ensure the affordability of much-needed global health technologies.
For instance, in many low-income countries, where budget resources are stretched thin, health systems may not be able to ensure the safety of health tools and products. This leads to situations where the very injections meant to prevent or treat disease can end up causing it instead because under-resourced, overburdened health care programs reuse or fail to properly sterilize contaminated injection equipment.
Recognizing the tremendous negative impact that unsafe injection practices can cause, PATH researched how to improve injection practices to reduce the risk of transmitting bloodborne diseases such as HIV and hepatitis B in low-resource settings. The US government, through the US Agency for International Development, provided funding to PATH, enabling us to develop and patent a design for an auto disable (AD) syringe that automatically locks after a single injection.
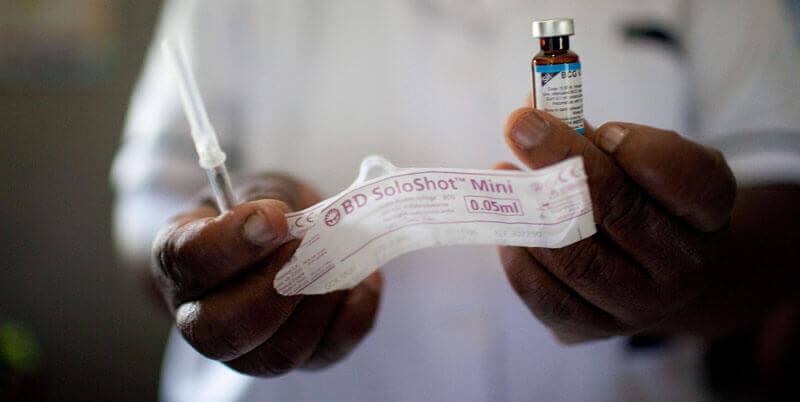
While PATH had the scientific and technical know-how to design the technology, a commercial partner was needed to validate the design, support large-scale production, and enable widespread distribution. PATH licensed the technology to Becton, Dickinson and Company (BD), the world’s largest syringe manufacturer. BD brought product development expertise to advance the technology beyond proof of concept, manufacturing capabilities to produce a global supply, and distribution channels to ensure the availability of the technology in the public and private sectors. The US government’s investment in the product’s development—harnessed many times over by BD’s own investment in the device—enabled PATH and BD to pursue an aggressive distribution program that led to SoloShot™, the world’s first commercially available AD syringe.
A major boost to the introduction and use of AD syringes occurred when the United Nations Children’s Fund and the GAVI Alliance began purchasing and distributing AD syringes for their programs. The US government also accelerated the adoption of the technology by aggregating demandthrough the US President’s Emergency Plan for AIDS Relief (PEPFAR) program. Because injection safety was one of PEPFAR’s top priorities, the US government procured more than 250 million AD syringes for 11 countries through the Making Medical Injections Safer project over its five-year life. Since 1992, more than six billion vaccinations have been delivered using SoloShot™ in more than 40 countries. Studies have shown that transmission of bloodborne diseases caused by dirty needles has been reduced by 90 percent in programs using some form of AD syringe.
Creating partnerships that harness the assets of the private sector and the public sector together is a powerful means to ensure impact, scale, and sustainability for the world’s most vulnerable populations. The partnerships help align the strengths and requirements of each sector to ensure that appropriate technologies and innovations reach communities and individuals with the greatest need.