Led US COVID-19 product development efforts for vaccines, diagnostics, and therapeutics, including advancing all therapeutics and first-generation vaccines approved or authorized by the US Food and Drug Administration (FDA).
Global Health R&D across the US government | BARDA
Global Health R&D at the Biomedical Advanced Research and Development Authority
 What does BARDA do for global health R&D?
What does BARDA do for global health R&D?
The Biomedical Advanced Research and Development Authority (BARDA) supports the development of vaccines, drugs, and other medical countermeasures (MCMs) to protect Americans against threats to public health, including emerging infectious diseases, pandemic influenza, and antimicrobial resistance (AMR).
 Why is BARDA’s role in global health R&D important?
Why is BARDA’s role in global health R&D important?
BARDA works with industry and other partners to bridge the “valley of death” between basic research and product development, where research and development (R&D) efforts most often fail. Through unique contracting and incentive mechanisms, BARDA’s partnerships ensure promising research is translated into urgently needed medical products by creating commercial incentives for developers that would otherwise not exist. During the COVID-19 pandemic, BARDA’s prominence and funding grew exponentially as the agency was charged with leading the US government’s MCM R&D portfolio, demonstrating how—with sufficient, sustained funding—BARDA is uniquely equipped to advance products against a range of other global health threats.
 Impact of Investment
Impact of Investment
BARDA support has helped advance at least:
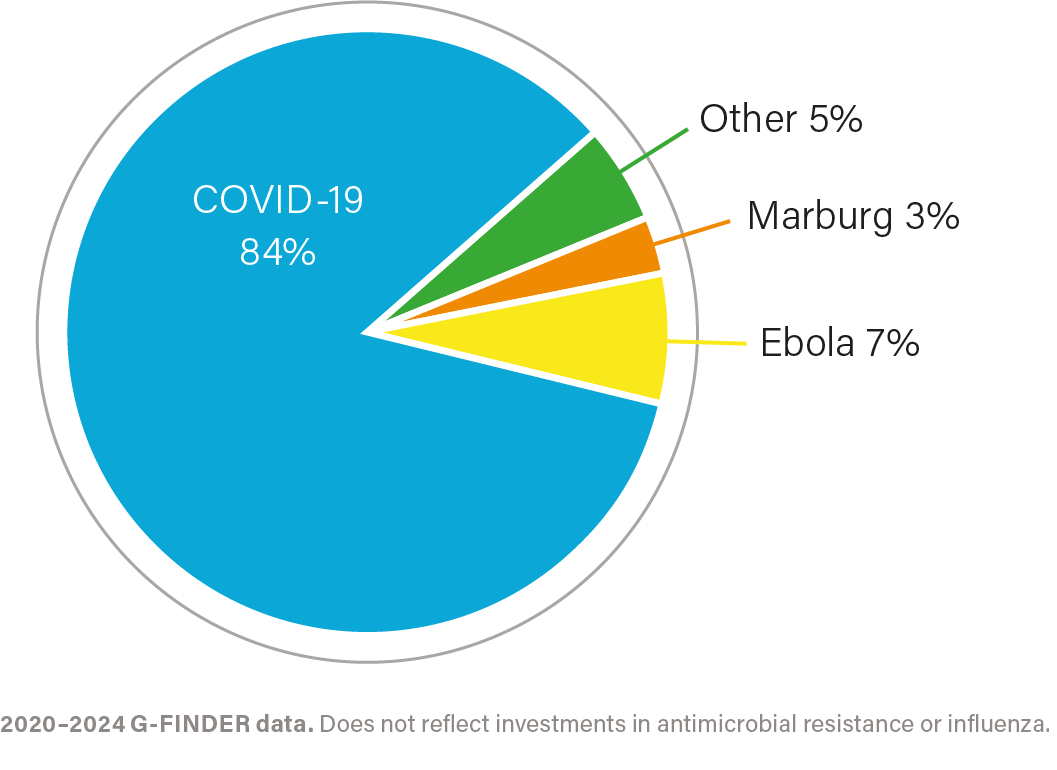
 R&D investment by health area, 2020–2024
R&D investment by health area, 2020–2024
R&D SUCCESS STORIES
COVID-19
EBOLA
Development of both Ebola Zaire vaccines, as well as two Ebola treatments and one rapid diagnostic test approved by FDA.
AMR
Accelerating antibacterial research through the Combating Antibiotic Resistant Bacteria Biopharmaceutical Accelerator, or CARB-X, public-private partnership, which has advanced 3 available diagnostics and supported 123 projects in 14 countries, to build the world’s largest early development pipeline of antibacterial innovations.
MPOX
Development of the one of the two FDA-approved vaccine for smallpox/mpox, which was deployed during the 2022–2023 outbreak, as well as a new FDA-approved freeze-dried formulation of the vaccine that has a longer shelf life.
ZIKA
Development of Zika diagnostics, including tests to identify infection and screen blood supplies.
INFLUENZA
Development of FDA-approved diagnostics, vaccines, therapeutics, and devices for seasonal and pandemic-risk influenza strains, alongside strengthening manufacturing capacity in low- and middle-income countries to enable rapid production of vaccines.
