Development of the first antiretroviral drugs and other subsequent therapies, which have collectively averted nearly 21 million AIDS-related deaths globally, and foundational science enabling a revolutionary new twice-yearly HIV prevention drug lenacapavir.
US agency series: Global health R&D across the US government
This six-part fact sheet series examines the contributions of US government agencies to advancing global health R&D. The series includes the National Institutes of Health, Department of State, Biomedical Advanced Research and Development Authority, Centers for Disease Control and Prevention, Department of War, and Food and Drug Administration.
Global Health R&D at the National Institutes of Health
 What does NIH do for global health R&D?
What does NIH do for global health R&D?
The National Institutes of Health (NIH) advances basic, applied, and clinical research across a range of global health areas and products. NIH facilitates this research through in-house programs and grants to universities, nonprofits, and other organizations and operates clinical trial networks that serve as the backbone for clinical trials taking place across America and the world.
 Why is NIH’s role in global health R&D important?
Why is NIH’s role in global health R&D important?
NIH is the United States’ leading medical research institution and a major contributor to global health research. Through the National Institute of Allergy and Infectious Diseases, NIH advances research on global infectious diseases; through the Eunice Kennedy Shriver National Institute of Child Health and Human Development, it supports maternal and child health research; through the Office of AIDS Research and Office of Research on Women’s Health, it coordinates cross-cutting HIV/AIDS research and women’s health research and inclusion policies; and through the Fogarty International Center, it strengthens international research capacity. Together this work underpins the development of new drugs, vaccines, diagnostics, and other tools that save and improve lives worldwide.
 Impact of Investment
Impact of Investment
NIH support has helped advance at least:
 R&D investment by health area, 2020–2024
R&D investment by health area, 2020–2024
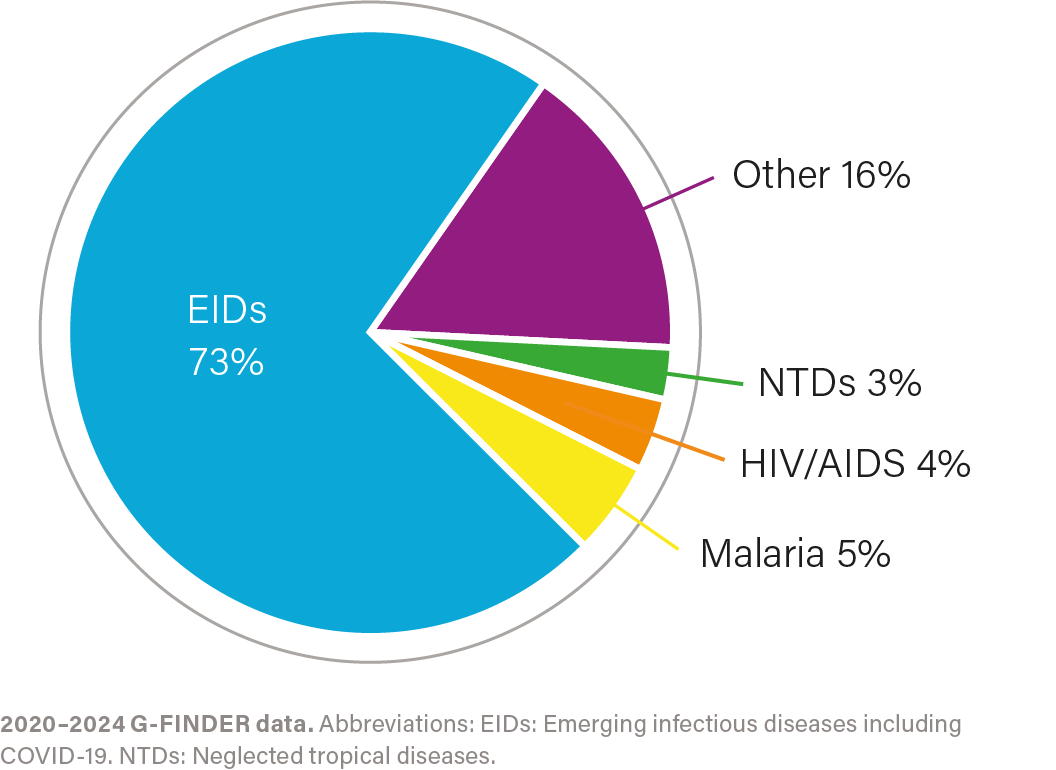
IMPACT OF INVESTMENT
HIV/AIDS
Development of the first antiretroviral drugs and other subsequent therapies, which have collectively averted nearly 21 million AIDS-related deaths globally, and foundational science enabling a revolutionary new twice-yearly HIV prevention drug lenacapavir.
TB
Development of a new drug for drug-resistant tuberculosis (TB), pretomanid, which is part of new regimens that has transformed treatment outcomes and reduced treatment time and could lead to 191,222 additional lives saved and $1.29 billion in global cost savings by 2034.
NTDs
Development of new tools to combat neglected tropical diseases (NTDs), including two treatments for visceral leishmaniasis, a single-dose oral treatment for sleeping sickness, a single-dose chikungunya vaccine, and rapid diagnostic tests for Chagas disease and river blindness.
CAPACITY-STRENGTHENING
The Fogarty International Center has provided research training to more than 8,500 US and foreign scientists working in low- and middle-income countries, including alumni who have played vital roles in the Ebola, Zika, COVID-19, and HIV/AIDS responses.
GLOBAL HEALTH SECURITY
Advancement of key discoveries that led to the development of the mRNA vaccine technology that was successfully leveraged for COVID-19 vaccines and is being advanced for other disease threats, and development of both licensed Ebola Zaire vaccines and both licensed treatments.
WOMEN'S HEALTH
Pioneered discovery that led to first commercially available vaccine for HPV, which has protected millions around the world from cervical cancer, and supported development of a year-long, reusable contraceptive vaginal ring and a new first-in-class antibiotic for gonorrhea, a disease which can cause infertility and pregnancy complications.
Global Health R&D at the Department of State
 What does the Department of State do for global health R&D?
What does the Department of State do for global health R&D?
The State Department invests in targeted efforts to advance the development of technologies for tuberculosis (TB) and emerging infectious diseases and supports the scale up and delivery of health products and services across a range of health areas through its global health programs. Through the new, congressionally directed Prevention, Treatment and Response Initiative and the department’s proposed Innovation Fund, the State Department has an opportunity to expand its role in research and development (R&D) by supporting innovations that accelerate progress in line with US bilateral agreements and partner-country priorities.
 Why is the Department of State’s role in global health R&D important?
Why is the Department of State’s role in global health R&D important?
Following the integration of the US Agency for International Development (USAID) into the State Department, the department is now the only US agency with a core mandate focused on global health and development, uniquely positioning it to support innovations that advance global health outcomes. For decades, USAID invested in the late-stage development of affordable, fit-for-purpose technologies designed for low-resource settings—addressing urgent needs often overlooked by other research agencies. As part of the integration, most former USAID-supported health R&D projects were terminated, creating a significant gap in US support for essential global health tools needed to address ongoing and emerging health challenges—one that State is well equipped to close.
 Impact of Investment
Impact of Investment
Former USAID support had helped advance at least:
 Current areas of Department of State R&D support
Current areas of Department of State R&D support
Health security: Oversees US contribution to the Coalition for Epidemic Preparedness Innovations [CEPI], a global partnership advancing vaccines for epidemic- and pandemic-prone diseases.
![]() TB: Supports select drug and diagnostic development projects, including research to develop shorter, more effective treatment regimens for drug-resistant TB.
TB: Supports select drug and diagnostic development projects, including research to develop shorter, more effective treatment regimens for drug-resistant TB.
Delivery and scale up Supports the delivery and scale up of existing and new health products. For example, the department is partnering with Gilead Sciences to rapidly scale up lenacapavir, a long-acting HIV prevention drug, in high-burden HIV countries.
OPPORTUNITY FOR STATE DEPARTMENT
The State Department has an opportunity to reinvigorate and build upon critical research formerly led by USAID, including:
HIV/AIDS
Advancing HIV vaccines, essential to ending the global pandemic, and new women-centered HIV prevention technologies, including on-demand inserts, implants, vaginal rings, and multipurpose prevention tools.
MALARIA
Developing next-generation malaria vaccines that overcome limitations of existing vaccines and improve efficacy, durability, and affordability, and continuing investment in new antimalarial drugs to stay ahead of emerging drug resistance.
VECTOR CONTROL
Developing new insecticides and innovative vector-control tools to combat mosquito-borne diseases—work that is increasingly critical as insecticide resistance threatens malaria control and elimination efforts worldwide.
NTDs
Advancing development of new medicines and evaluation of new and existing diagnostic tools for priority neglected tropical diseases (NTDs) that cause substantial illness, disability, and economic burden in low-resource communities worldwide.
WOMEN'S HEALTH
Advancing long-acting, user-controlled contraceptive innovations that better meet the needs of women worldwide with limited access to routine health care, including a long-acting, biodegradable implant and a contraceptive patch.
TB
Developing more effective TB vaccines, given the limited protection provided by the only existing vaccine in adults and adolescent, and adapting and evaluating new drug regimens for children with drug-resistant TB, who remain underserved by current options.
FORMER USAID R&D SUCCESS STORIES
HIV/AIDS
Development of a woman-controlled microbicide vaginal ring, the first long-acting HIV prevention method to receive a positive regulatory opinion.
MALARIA
Development of the world’s first malaria vaccine and a single-dose treatment for relapsing malaria, which simplifies care and reduces non-adherence that can contribute to drug resistance.
TB
Development of child-friendly TB medicines now adopted in more than 123 countries, representing over 75 percent of the global childhood TB burden.
MATERNAL HEALTH
Development of a low-cost, easy-to-use uterine device that applies pressure to slow excessive bleeding after childbirth, which is the leading cause of maternal mortality worldwide.
MENINGITIS
Development of a low-cost meningitis A vaccine, which, as of 2023, has been delivered to more than 350 million people in 24 countries, virtually eliminating meningitis A wherever it has been used.
NTDs
Development of the first oral drug and a point-of-care diagnostic test for lymphatic filariasis, a parasitic disease that can cause lifelong swelling, pain, and disability.
IMPACT OF INVESTMENT
Global Health R&D at the Biomedical Advanced Research and Development Authority
 What does BARDA do for global health R&D?
What does BARDA do for global health R&D?
The Biomedical Advanced Research and Development Authority (BARDA) supports the development of vaccines, drugs, and other medical countermeasures (MCMs) to protect Americans against threats to public health, including emerging infectious diseases, pandemic influenza, and antimicrobial resistance (AMR).
 Why is BARDA’s role in global health R&D important?
Why is BARDA’s role in global health R&D important?
BARDA works with industry and other partners to bridge the “valley of death” between basic research and product development, where research and development (R&D) efforts most often fail. Through unique contracting and incentive mechanisms, BARDA’s partnerships ensure promising research is translated into urgently needed medical products by creating commercial incentives for developers that would otherwise not exist. During the COVID-19 pandemic, BARDA’s prominence and funding grew exponentially as the agency was charged with leading the US government’s MCM R&D portfolio, demonstrating how—with sufficient, sustained funding—BARDA is uniquely equipped to advance products against a range of other global health threats.
 Impact of Investment
Impact of Investment
BARDA support has helped advance at least:
 R&D investment by health area, 2020–2024
R&D investment by health area, 2020–2024
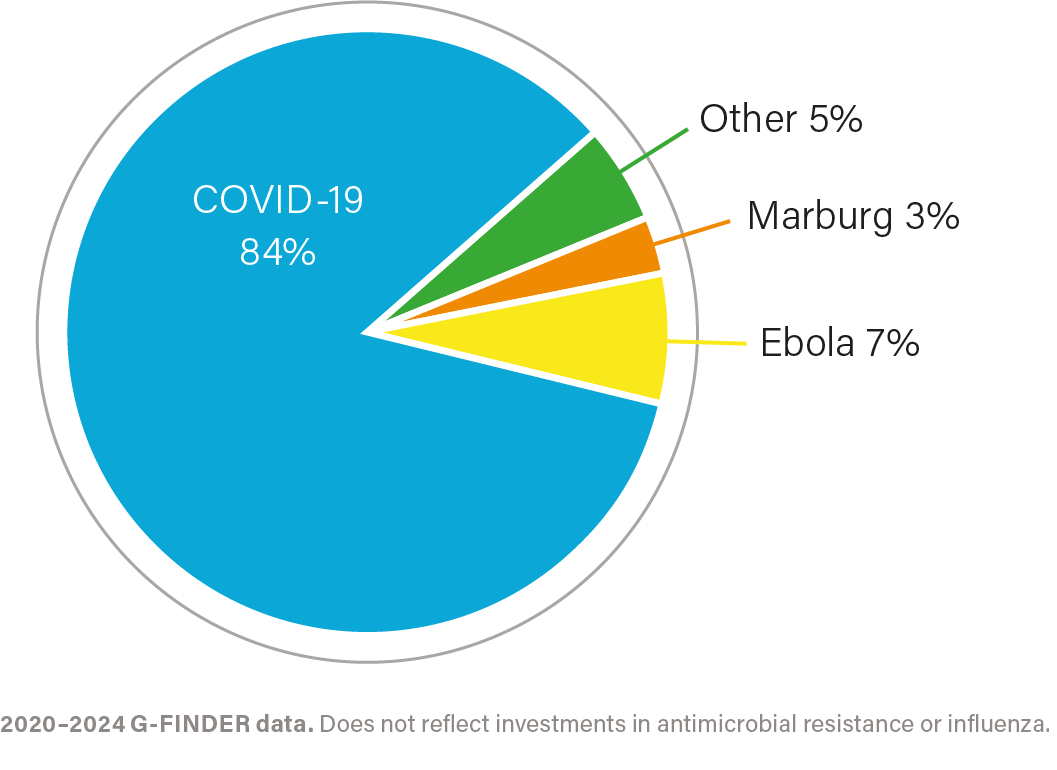
IMPACT OF INVESTMENT
COVID-19
Led US COVID-19 product development efforts for vaccines, diagnostics, and therapeutics, including advancing all therapeutics and first-generation vaccines approved or authorized by the US Food and Drug Administration (FDA).
EBOLA
Development of both Ebola Zaire vaccines, as well as two Ebola treatments and one rapid diagnostic test approved by FDA.
AMR
Accelerating antibacterial research through the Combating Antibiotic Resistant Bacteria Biopharmaceutical Accelerator, or CARB-X, public-private partnership, which has advanced 3 available diagnostics and supported 123 projects in 14 countries, to build the world’s largest early development pipeline of antibacterial innovations.
MPOX
Development of the one of the two FDA-approved vaccine for smallpox/mpox, which was deployed during the 2022–2023 outbreak, as well as a new FDA-approved freeze-dried formulation of the vaccine that has a longer shelf life.
ZIKA
Development of Zika diagnostics, including tests to identify infection and screen blood supplies.
INFLUENZA
Development of FDA-approved diagnostics, vaccines, therapeutics, and devices for seasonal and pandemic-risk influenza strains, alongside strengthening manufacturing capacity in low- and middle-income countries to enable rapid production of vaccines.
Global Health R&D at the US Centers for Disease Control and Prevention
 What does CDC do for global health R&D?
What does CDC do for global health R&D?
The US Centers for Disease Control and Prevention (CDC) protects people at home and abroad through disease surveillance, rapid outbreak response, and research and development (R&D) of diagnostics, drugs, and other technologies to combat infectious diseases. Not only does CDC’s research advance new diagnostic, prevention, and surveillance technologies, it also evaluates the effectiveness of existing tools to inform their use and future R&D needs. CDC’s Global Health Center, National Center for Emerging and Zoonotic Infectious Diseases, and National Center for HIV/AIDS, Viral Hepatitis, STD, and Tuberculosis Prevention lead much of the agency’s global health R&D work.
 Why is CDC’s role in global health R&D important?
Why is CDC’s role in global health R&D important?
CDC has unique expertise and capacity to detect, track, and contain infectious disease outbreaks and develop the right technologies to advance these efforts. CDC’s work is critical to protecting Americans and people around the world from emerging epidemics, as well as monitoring the impact of current tools and global health programs to maximize investment.
 Impact of Investment
Impact of Investment
CDC support has helped advance at least:
 R&D investment by health area, 2020–2024
R&D investment by health area, 2020–2024
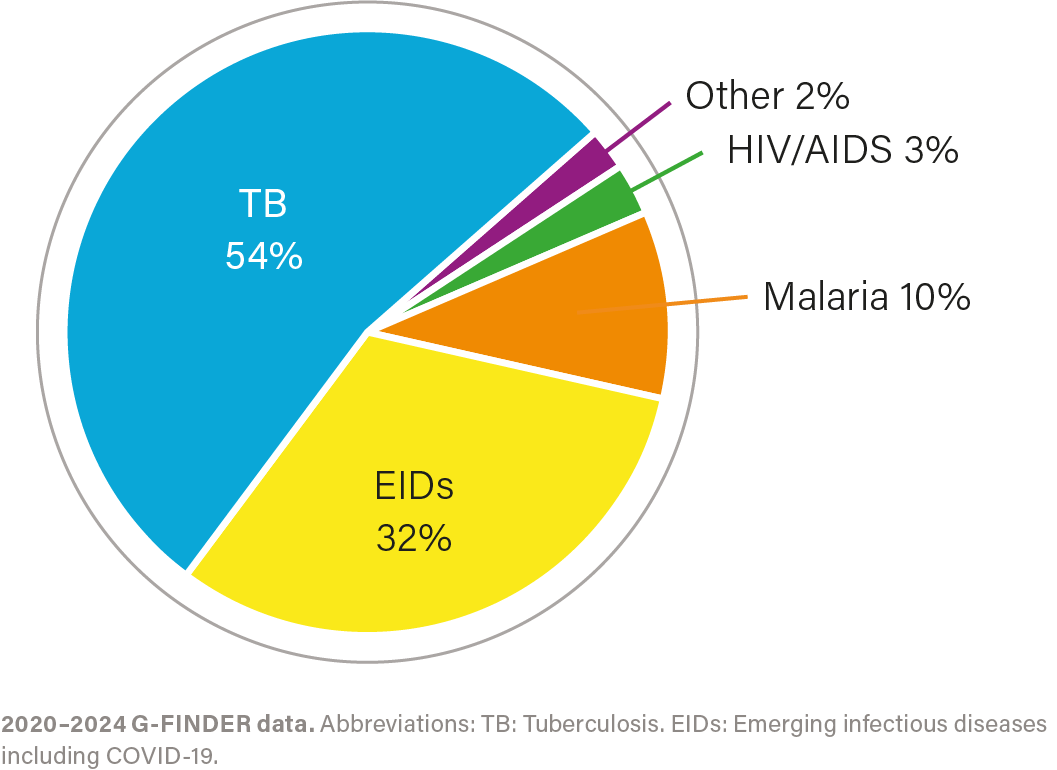
IMPACT OF INVESTMENT
MALARIA
Co-development of the world’s first malaria vaccine and creation of an innovative, easy-to-use tool to rapidly identify a malaria-transmitting mosquito species without need for sophisticated laboratory infrastructure or specialized training.
TB
Development of simplified, better-tolerated tuberculosis (TB) treatments through the Tuberculosis Trials Consortium, which have dramatically reduced TB treatment times and costs.
EBOLA
Development, evaluation, and distribution of rapid diagnostics for Ebola during the 2014–2015 outbreak, including a test that can provide results in as little as four minutes, as well as support for the clinical trials of the world’s first approved Ebola vaccine.
VECTOR CONTROL
Evaluation of insecticide-treated bednet durability to maximize impact and cost-effectiveness, and advancement of an innovative mosquito control approach that releases mosquitoes infected with a naturally occurring bacterium, passed to offspring, that blocks dengue and other viruses, reducing transmission across generations.
NTDs
Development of improved diagnostic tools for neglected tropical diseases (NTDs), including new tests for dengue, elephantiasis, river blindness, Guinea worm, and schistosomiasis.
CAPACITY-STRENGTHENING
Training of more than 20,500 disease detectives in more than 80 countries through its flagship global Field Epidemiology Training Program.
Global Health R&D at the Department of War
 What does the DoW do for global health R&D?
What does the DoW do for global health R&D?
The Department of War (DoW) supports research and development (R&D) for infectious diseases, antimicrobial resistance, and other health conditions that pose a risk to US national security and service members stationed abroad.
 Why is the DoW’s role in global health R&D important?
Why is the DoW’s role in global health R&D important?
While DoW research first and foremost aims to protect service members while overseas, it also helps generate vaccines, drugs, and other health tools to combat diseases that are endemic in the world’s poorest places. Additionally, because DoW focuses on producing health tools for austere settings like the battlefield, the tools it advances are often well-suited for use in low-resource communities worldwide.
DoW research is unique in spanning across all stages of R&D, from basic research to late-stage clinical development and manufacturing, making it the only US agency that can single-handedly advance a single technology from early research to end-stage product.
 Impact of investment
Impact of investment
DoW support has helped advance at least:
 R&D investment by health area, 2020–2024
R&D investment by health area, 2020–2024
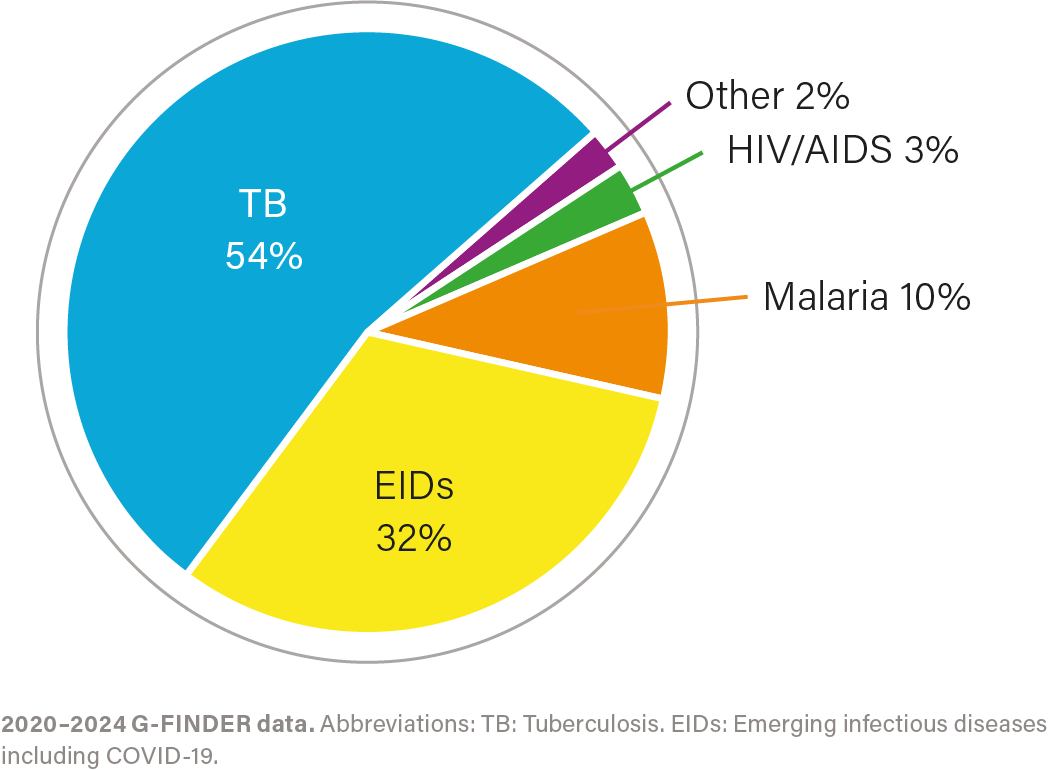
IMPACT OF INVESTMENT
MALARIA
Development of nearly every antimalarial drug approved by the US Food and Drug Administration (FDA) and the world’s first approved malaria vaccine, which alongside the second approved vaccine, have reached 24 million doses delivered across 20 countries.
HIV/AIDS
Led first-ever HIV trial to show efficacy in 2009 and supports ongoing development of potential HIV/AIDS vaccine regimens, including a trial in Uganda that combines experimental vaccines, an Army-developed adjuvant, and a novel dose escalation approach in hopes of boosting immune response.
DIARRHEAL DISEASES
Ongoing development of affordable vaccines against Enterotoxigenic Escherichia coli and Shigella, and creation of the technology used in a water chlorinator device, which gives low-resource communities access to safe, affordable drinking water.
EBOLA
Development of both Ebola Zaire vaccines, which have been used to successfully quell recent outbreaks of the disease, and a rapid, automated diagnostic test.
COVID-19
Co-developed a rapid test for cutaneous leishmaniasis, a parasitic skin infection transmitted by sandflies, and continues to support surveillance and research efforts by operating the only accredited leishmaniasis reference diagnostic laboratory in North America and hosting the largest sand fly repository in the world.
CAPACITY-STRENGTHENING
Operates a network of overseas labs and medical research facilities, which provides bases for infectious disease R&D, disease surveillance, and capacity-strengthening in partner nations.
Global Health R&D at the US Food and Drug Administration
 What does FDA do for global health R&D?
What does FDA do for global health R&D?
The US Food and Drug Administration (FDA) regulates the safety and efficacy of drugs, vaccines, and other medical products marketed in the United States, which can include products also designed for use overseas. FDA also works with international organizations and regulators in low- and middle-income countries (LMICs) to strengthen regulatory capacity and provide technical assistance.
 Why is FDA’s role in global health R&D important?
Why is FDA’s role in global health R&D important?
FDA approval of a product serves as a “gold standard” that can expedite regulatory review in LMICs. This effect, combined with the agency’s work in regulatory capacity-strengthening, helps ensure new global health technologies are safe, effective, and accessible in low-resource settings.
 Ensuring safe & effective products
Ensuring safe & effective products
FDA has approved more than 165 drugs, vaccines, and diagnostics for neglected and emerging diseases.
FDA has 217 formal arrangements for information sharing and technical assistance with regulatory authorities in 55 countries.
Contributions to global health R&D

IMPACT OF INVESTMENT
CAPACITY-STRENGTHENING
Development of partnerships to strengthen regulatory capacity in LMICs, including collaborations with the Pan American Network for Drug Regulatory Harmonization to share expertise and support capacity-strengthening.
HIV/AIDS
Creation of a “tentative approval” process to certify antiretroviral (ARV) drugs that the President’s Emergency Plan for AIDS Relief, or PEPFAR, purchases for use outside the United States. Through the program and standard review, FDA has approved more than 250 ARVs, which have helped support treatment for more than 20.5 million people worldwide via PEPFAR.
HEALTH EMERGENCIES
Granted emergency use authorization for more than 400 medical products for COVID-19, 8 products for mpox, 18 diagnostics for Zika, and 11 diagnostics for Ebola, facilitating their use during these crises.
MENINGITIS
Development of critical technology used in a low-cost meningitis A vaccine, which has been delivered to an estimated 360 million people across sub-Saharan Africa since 2010, virtually eliminating meningitis A wherever it has been used.
NTDs
Release of guidance documents to aid organizations in developing drugs for neglected tropical diseases (NTDs) and issuance of 14 priority review vouchers (PRVs) as part of the NTD PRV program intended to stimulate private-sector investment in NTD R&D.
TB
Use of an accelerated approval pathway to speed review and approval of two new drugs to treat drug-resistant tuberculosis (TB).
