Joanna BreitsteinTB Alliance
Joanna Breitstein is the Communications Director at TB Alliance, a not-for-profit organization dedicated to finding faster-acting and affordable drug regimens to fight TB.
In this guest post, Joanna Breitstein, director of communications at the TB Alliance, highlights the response to a new study that indicates a novel tuberculosis (TB) combination drug may be on the horizon.
Global press coverage this week cites a Lancet study suggesting that scientists have made significant progress toward developing an effective and affordable treatment for TB, a disease that every year kills more than 1.4 million people and is the primary cause of death among people with AIDS.
Released on Monday during a presentation at the International AIDS Conference (IAC), the results of the Phase II trial in South Africa offer hope for a treatment breakthrough amid the growing and dangerous epidemic of drug-resistant forms of TB that, in some cases, are becoming untreatable.
In a story by the Associated Press, science writer Lauran Neergaard reports, “Scientists are hot on the trail of a new tuberculosis treatment that a small study suggests might one day offer an alternative to battle this deadly lung disease, even if it’s resistant to today’s two main drugs.”
Asked to comment on the new study, IAC Co-Chair Dr. Diane Havlir of the University of California,San Francisco told the Associated Press, “We are cautiously optimistic that we are at the dawn of a new era for TB drugs."
In The Guardian, Sarah Boseley quotes TB Alliance CEO Dr. Mel Spigelman, who calls the implications of the findings “huge.”
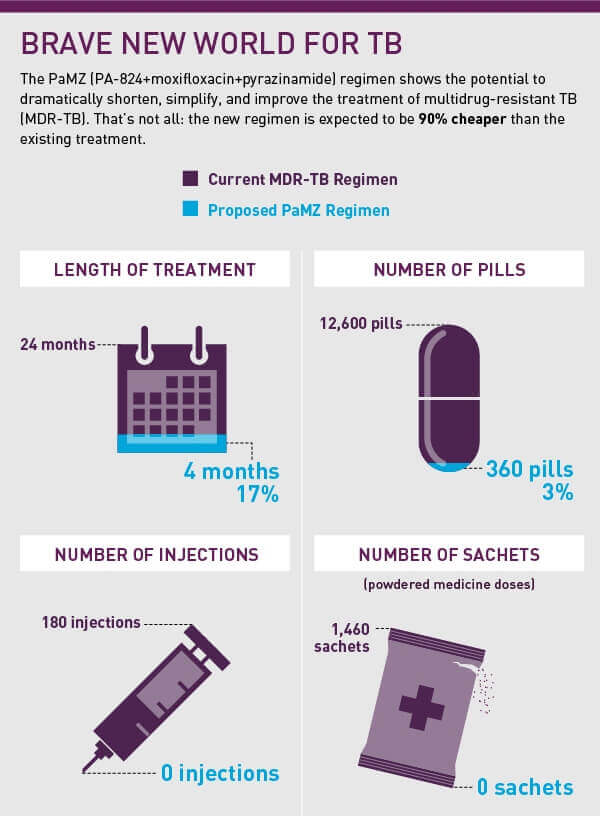
"People with drug-susceptible and [some forms of] drug-resistant TB could be cured in as little as four months or even shorter,” Spigelman tells Boseley. “In the case of multi-drug-resistant TB, we could be reducing their treatment by one to two years or more. The regimen also promises to be in the order of 90 percent cheaper than the current [multi-drug resistant] treatment regimen. There is potential for a dramatically shorter, simpler, cheaper, and more effective regimen all at the same time.’”
Also quoted in The Guardian, principal investigator Dr. Andreas Diacon of Stellenbosch University in South Africa says he speaks as a clinician and researcher. "Treating drug-sensitive and drug-resistant TB with the same regimen can simplify the delivery of TB treatment worldwide. The results of this study give healthcare providers on the front lines of the TB epidemic hopes for better, faster tools needed to stop this disease."
Spigelman notes in Amy Maxmen’s article in Nature that the Lancet study also validates a new model for speeding development of such treatments. “After a new drug reaches the market, the traditional route is to work from the standard of care and substitute one drug for another. But if you do it in an incremental way, that will give you incrementally better results,” Spigelman says. “Our goal is to shorten the path to getting a new, cohesive regimen out there in one fell swoop.”
Mario Raviglione, director of the Stop TB Department at the World Health Organization, says testing multiple new TB drug candidates simultaneously has already proven to be a major advance.
"Because of testing drugs in combination, we have already saved several years in the research process to find new, effective regimens to treat TB," according to Raviglione. "The results look strongly promising from this early trial. If further testing holds up these results and the regimen is affordable in poor countries, it is huge progress. We could shorten drug regimens substantially for everyone, regardless of whether the form of TB is sensitive or multi-drug resistant. That would be a dramatic step forward."
“If full clinical trials confirm its promise, the therapy could prove a boon for HIV-infected people,” writes Maxmen in Nature. “TB, which is common in its latent form in many parts of the developing world, often becomes active in those who are HIV-positive because of their weakened immune systems.”
Unlike some existing therapies, Maxmen continues, the “new combination does not seem to interact adversely with drugs for HIV.” To illustrate the situation on the ground, where both diseases wreak havoc, Maxmen quotes John Farley, deputy director of the antimicrobial product division at the US Food and Drug Administration who once worked with HIV patients in West Africa:
“…there were times when our HIV patients would die of drug-resistant TB before we could even get them their antiretroviral therapy,” says Farley in Maxmen’s story.