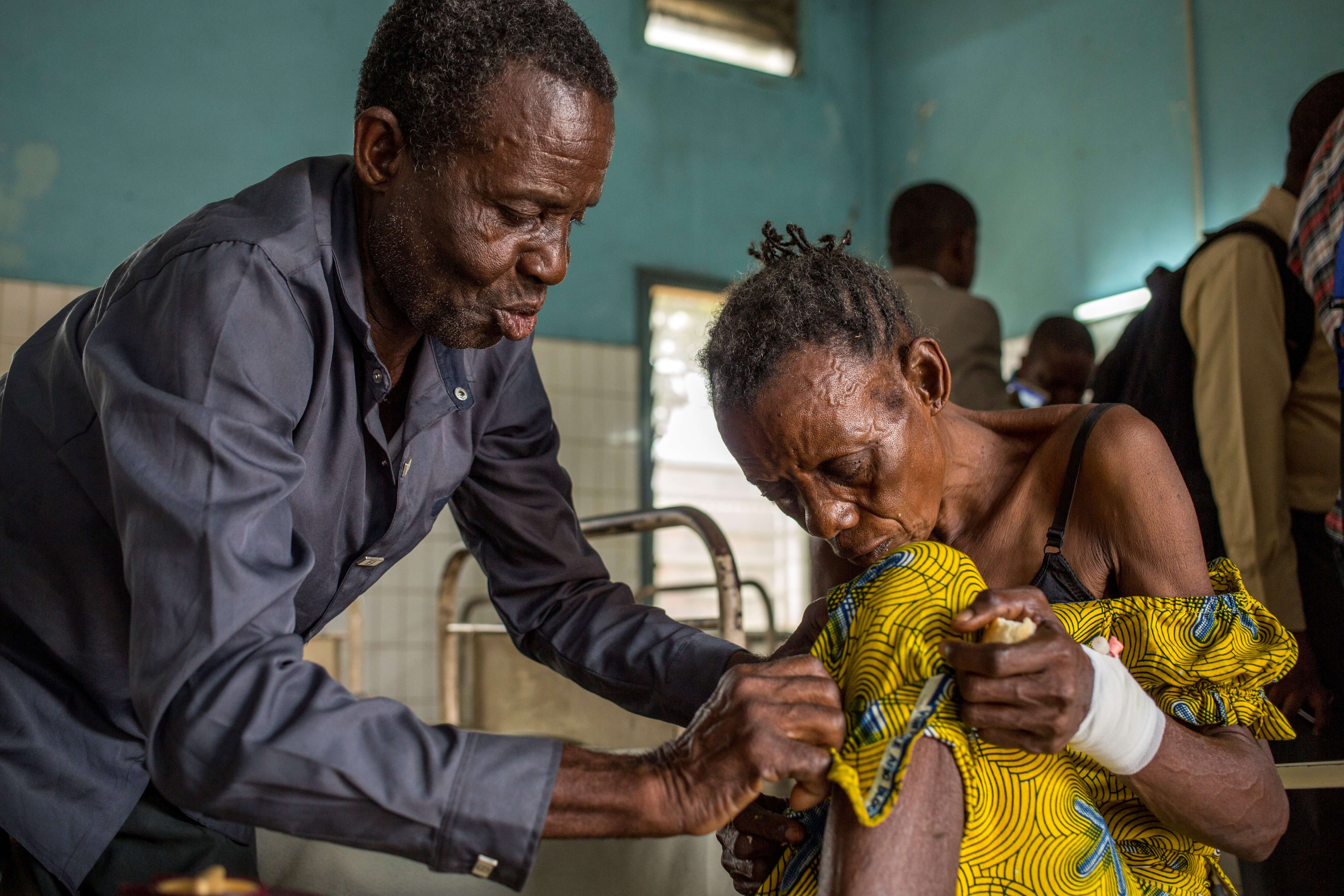Two thousand eighteen was a turbulent year in global health. Between reemerging crises, breakthrough scientific discoveries, and the continued looming
threat of health research funding cuts, 2018 took us on a roller coaster ride of peril and promise. Now as the year comes to an end, GHTC is taking
a moment to reflect on the global health innovation stories and news that shaped 2018.
Here are the highlights from our year in review:
1. A long-neglected disease sees a breakthrough
 A patient suffering from sleeping sickness visits a hospital in DRC. PATH/Georgina Goodwin
A patient suffering from sleeping sickness visits a hospital in DRC. PATH/Georgina Goodwin
In 2018, a revolutionary treatment emerged for a neglected tropical disease, sleeping sickness. The first all-oral drug for sleeping sickness, developed by the Drugs for Neglected Diseases initiative (DNDi) and Sanofi,
received a positive scientific opinion from the European Medicines Agency—paving the way for the treatment to be rolled out in endemic countries
in 2019. This groundbreaking treatment, known as fexinidazole, is a tablet taken once daily for ten days that treats the most common form of sleeping sickness, a disease spread by tsetse flies, which causes neuropsychiatric symptoms and can be deadly if left untreated.
Current treatments for sleeping sickness require a complicated series of infusions and pills, are costly, and involve long hospital stays. Fexinidazole
is a simple once-a-day pill that could reduce the logistical burdens to treating a disease for which approximately 65 million people in sub-Saharan
Africa are at risk.
2. TB captures the spotlight as new research demonstrates promise
The world’s largest infectious disease killer finally received some well-deserved attention this year as world leaders convened the first-ever United Nations
High-Level Meeting on Tuberculosis, at which member states committed to closing the US$1.3 billion annual gap in funding for tuberculosis (TB) research.
While time will tell whether this moment will prove a powerful catalyst or an
empty promise, the science continues to advance as we wait for the politics to play catch up.
This year we saw several exciting new developments in TB research. Aeras and GlaxoSmithKline released the results of a phase 2b clinical trial that showed their candidate vaccine prevented TB from developing in half of those who received
it. If it succeeds in additional testing, this vaccine would represent an improvement over the existing vaccine in use and would be the first new TB
vaccine developed in over a century. On the treatment side, TB Alliance and partners launched the
SimpliciTB trial, to test a new all-oral drug regimen for treating both drug-sensitive and drug-resistant TB, known as BPaMZ, which
could slash treatment times for TB by one-third or more and is projected to be markedly less expensive than current therapy. Additionally, TB Alliance
was honored this year by GHTC with an
Innovating for Impact Partnership Award for the development of child-friendly TB medicines, which are now available in 86 countries and counting.
3. A single dose cure for malaria is approved
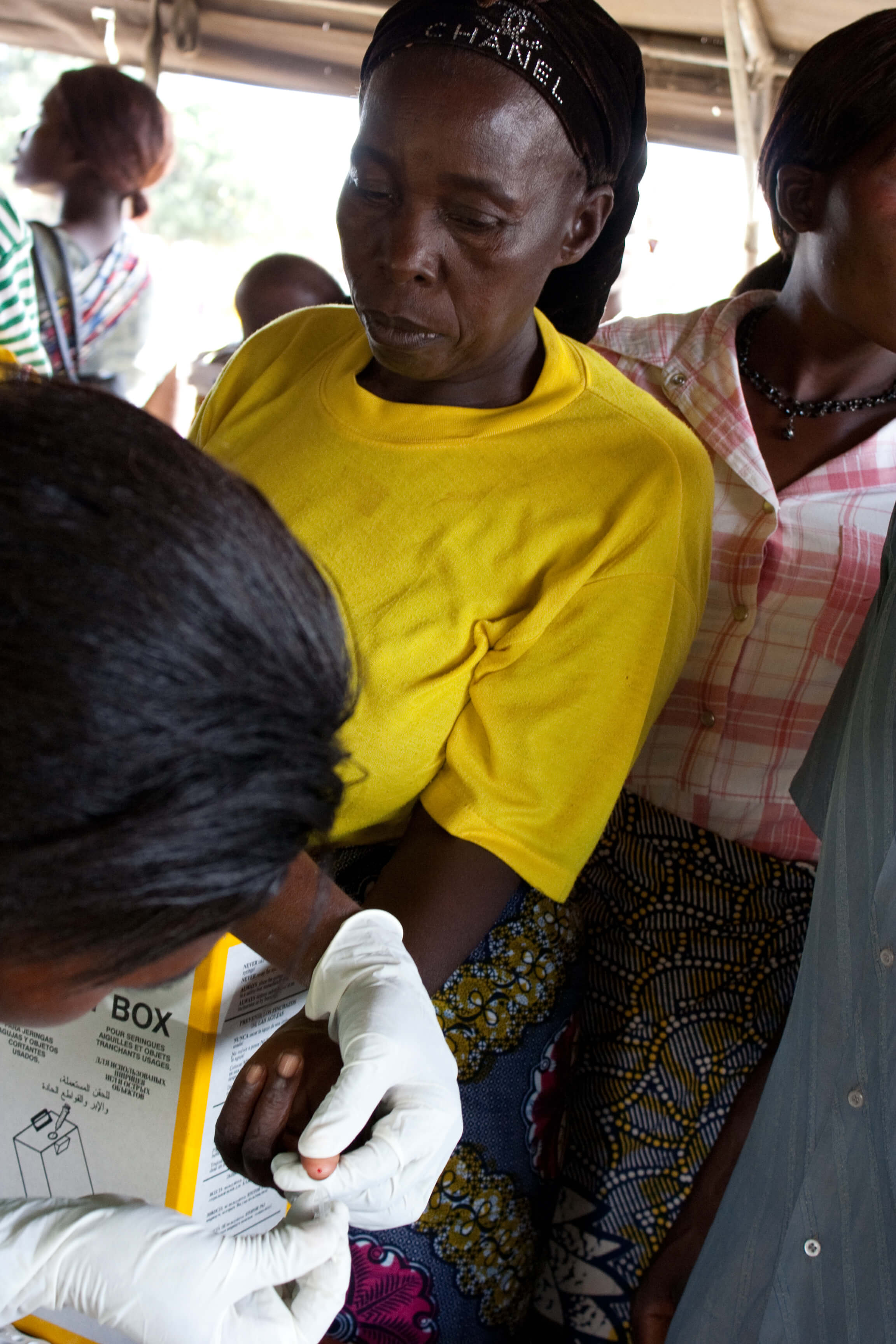 A patient in Zambia receives a malaria diagnostic test. PATH/Gena Morgan
A patient in Zambia receives a malaria diagnostic test. PATH/Gena Morgan
This year, the global health community witnessed regulatory approval of the first new treatment for Plasmodium vivax malaria in 60 years by the US Food and Drug Administration (FDA). The new drug,
tafenoquine, is a single-dose
treatment developed by GlaxoSmithKline and Medicines for Malaria Venture. P. vivax malaria causes around 8.5 million infections every year in Asia,
Latin America, and parts of Africa. It’s tricky to combat because the P. vivax parasite can lie dormant in the liver, where it reactivates,
and can cause new episodes of malaria weeks, months, or even years after the initial infection. In a study of the treatment, 60 percent saw no relapse
six months after taking the drug. Tafenoquine has been submitted for approval to the Australian regulatory authority and is slated to be submitted
for approval in malaria-endemic countries. Experts hope this new treatment will accelerate the path to elimination of this mosquito-borne disease.
4. Innovations show promise while the battle against Ebola rages on
One of the most disheartening stories of 2018 was the reemergence of Ebola in the Democratic Republic of Congo (DRC). Ongoing conflict in the region has
hampered response efforts, with the outbreak now reaching 500 confirmed cases. Yet, amidst this crisis, there is one reason for optimism: An experimental
vaccine appears to be helping those it reaches. Close to 45,000 people have received the experimental rVSV-ZEBOV Ebola vaccine—the largest use
of the product since a clinical trial in 2015 showed it worked. While the vaccine’s effectiveness during this outbreak has not been formally assessed,
World Health Organization (WHO) officials believe it is having a major impact. The vaccine’s developer, Merck, announced last month that it has begun the process of submitting evidence to seek FDA approval, a crucial step to bring the vaccine to market. Meanwhile, new preliminary research also suggests experimental treatments deployed during the outbreak are improving survival rates, and researchers
have begun clinical trials in eastern
DRC to test the effectiveness of four of these experimental drugs. This research raises new hope that science may soon offer us both a vaccine and
treatments to combat Ebola.
5. Congress pushes back against cuts to global health and medical research
While the Trump Administration proposed in its fiscal year (FY) 2018 and 2019 budgets to slash funding for global health and medical research programs,
in the end Congress pushed back. In March, Congress passed the FY18 omnibus, which protected, and in many cases increased, funding to key US agencies and programs that advance global health
research and development (R&D). In September, Congress passed an FY19 spending bill covering the Defense, Labor, Health and Human Services (LHHS), and Education departments. The bill boosted funding
for medical research across the Department of Health and Human Services, with the National Institutes of Health receiving a $2 billion increase and
the US Centers for Disease Control and Prevention and the Biomedical Advanced Research and Development Authority also receiving modest increases.
These funding bills represent a forceful rebuke by Congress of the Administration’s proposals to draw back support for global health and medical research
programs and demonstrate that Congressional policymakers on both sides of the aisle want to see the US continue its leadership in advancing innovation
to improve the health of those in need around the world. Given that funding for the US Agency for International Development (USAID) was maintained
through a continuing resolution that expires on December 21, we now await further action by Congress to determine how FY19 funding for global health
accounts at USAID will shake out.
6. New vaccines give us a better shot at saving lives
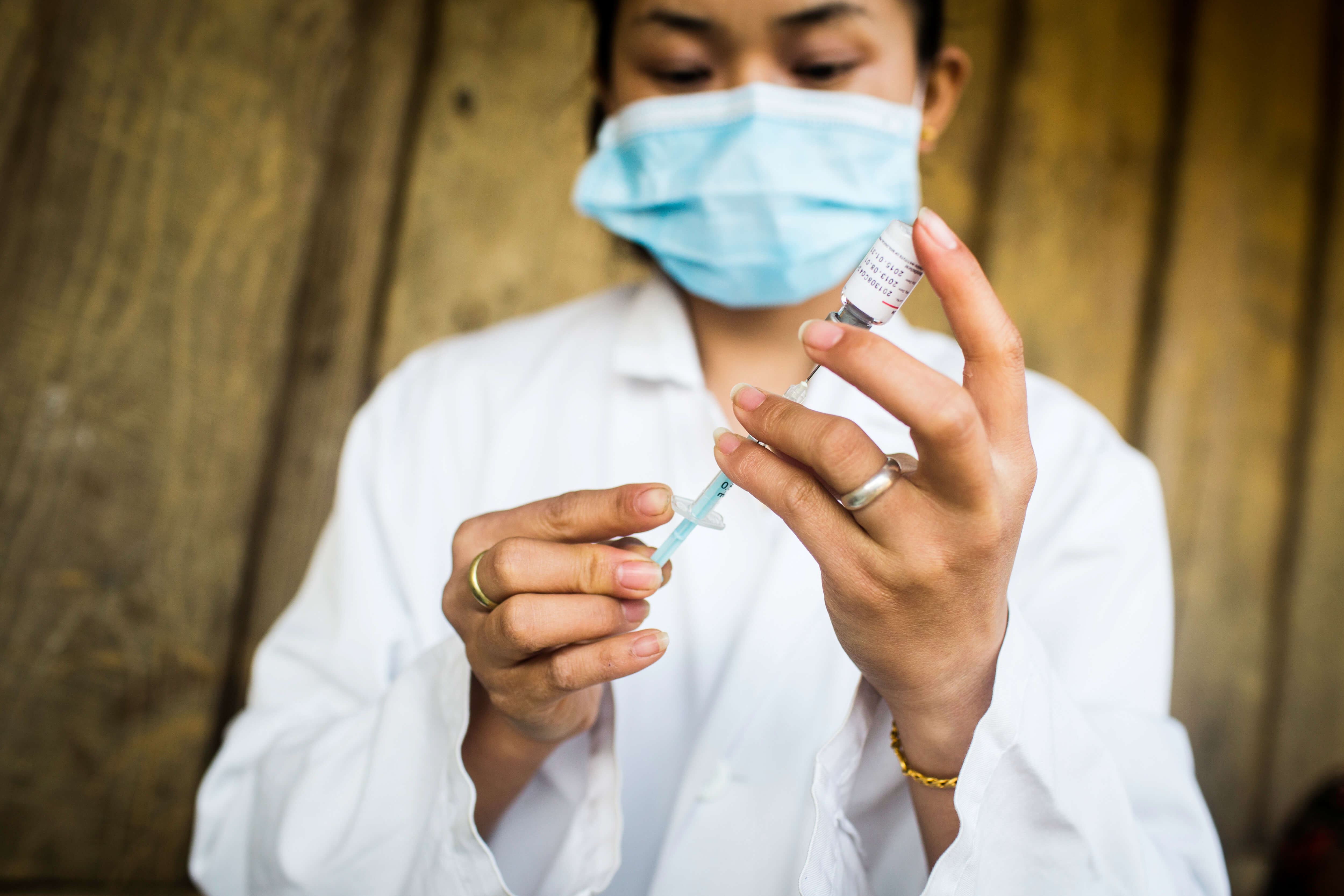 A healthworker in Laos prepares a vaccine. PATH/Aaron Joel Santos
A healthworker in Laos prepares a vaccine. PATH/Aaron Joel Santos
Two thousand eighteen was a year of innovations in vaccines. In what could be the first new polio vaccine in more than 50 years, researchers have developed
a way to
freeze-dry an inactivated poliovirus vaccine (IPV) so that it can be stored at higher temperatures and reconstituted for use without
losing its potency—potentially offering a new tool to help eradicate the disease. In other vaccine news, Johnson & Johnson (J&J) unveiled
promising early-stage results for a tetravalent mosaic-based HIV preventative vaccine regimen. Currently in large-scale clinical trials in sub-Saharan Africa, the vaccine marks
only the fifth time in history an HIV vaccine candidate has advanced to human clinical trials, offering new hope for a breakthrough in HIV prevention.
Additionally, WHO prequalified new vaccines to combat typhoid and rotavirus, which is the most common cause of severe diarrheal disease in children
worldwide. In January, WHO approved a new, highly effective and low-cost
typhoid vaccine for global use. In the same month, it prequalified
Rotavac, an oral rotavirus vaccine, and later in the year, approved
Rotasiil, a first-of-its-kind thermostable vaccine to prevent rotavirus.
7. Innovators strive to put greater choice in the hands of patients
One of the exciting themes we saw across so many global health stories this year was the power of innovation to put greater choice in the hands of patients.
People have unique needs, and our toolbox of global health technologies must reflect that. Several scientific developments this year promise to expand
family planning choices for women and their families. The first-of-its-kind, self-insertable and removable
one-year contraceptive vaginal ring received regulatory approval from the FDA;
new research from Uganda showed a self-injectable form of contraception can help women overcome barriers to consistent use; and
the first clinical trial was launched for a
male contraceptive gel,
a technology that could bring more equality to the burden of family planning.
8. Scientists pursue wild and weird ways to fight deadly diseases
 A dog in India. PATH/Gabe Bienczycki
A dog in India. PATH/Gabe BienczyckiWinston Churchill once said, “No idea is so outlandish that it should not be considered.” That’s clearly a motto researchers have taken to heart, as evidenced
by the number of stories we read this year about unusual and creative ways scientists are trying to fight infectious diseases. Here’s a sample of some
of our favorites. One could say it’s a bacteria-eat-bacteria world out there, with researchers studying whether
germ-eating microbes could be used to combat drug-resistant infections. Man’s best friend may also be man’s diagnostic tool, according
to new research showing that
dogs can be trained to detect malaria in people.
In the future, administering a vaccine could be as easy and pain-free as putting on a Band-Aid if
microneedle patches in development come to fruition. The contents of your takeout dinner might be the next frontier in HIV prevention,
as scientists have developed a technique to
modify a strain of rice to produce HIV-neutralizing proteins.
While these ideas may seem fantastical, history has taught us that groundbreaking discoveries can come from unusual places.
For other fascinating global health news delivered to your inbox weekly and to stay abreast of developments in 2019 and beyond, sign-up now to receive GHTC’s R&D News Roundup newsletter.