What do an onsite chlorine generator, fingerprint scanning technology for newborns, and temperature-monitoring wristband all have in common? The potential
to save the lives of mothers and newborns in some of the poorest, hardest-to-reach communities around the world.
These were among the many innovations pitched on July 27 at the DevelopmentXChange—the final stage of competition in the seventh annual Saving Lives at Birth: A Grand Challenge for Development,
a prize program sponsored by the US Agency of International Development and other partners to identify and advance transformative, cost-effective solutions
to improve maternal and newborn health, particularly during the first 48 hours after birth. Through the challenge, innovators compete for grants to
develop, test, and bring their ideas to scale.
This year, the challenge received more than 550 applications from across the world, with 53 teams chosen as finalists to compete at this year’s DevelopmentXChange.
At the conclusion of the event, 15 winners were selected, with additional grant recipients to be announced later this year in the transition-to-scale category.
GHTC members featured among finalists
Among the 53 finalists were five promising innovations from GHTC member organizations, profiled below:
Validation of an innovative, onsite, easy-to-use chlorine generator for infection prevention and control in labor wards*
PATH
 The chlorine generator for infection prevention and control in labor wards. (Photo: GHTC/Marissa Chmiola)With just salt, water, and a power source, a hospital or clinic can create chlorinated water onsite to disinfect equipment and surfaces to keep mothers
and babies healthy. That’s the idea behind the SE Flow Chlorinator—an easy-to-use, cost-effective chlorine generator designed for low-resource
settings.
The chlorine generator for infection prevention and control in labor wards. (Photo: GHTC/Marissa Chmiola)With just salt, water, and a power source, a hospital or clinic can create chlorinated water onsite to disinfect equipment and surfaces to keep mothers
and babies healthy. That’s the idea behind the SE Flow Chlorinator—an easy-to-use, cost-effective chlorine generator designed for low-resource
settings.
Infections are a leading cause of death for mothers and newborns during childbirth. While chlorine is one of the most widely-used and accepted methods
for disinfecting surfaces, getting chlorine to remote health care facilities can be a challenge due to combination of weak supply chains, inconsistent
transportation networks, and bureaucratic hurdles. The result: Close to one-third of facilities in low-income countries recently surveyed by the World
Health Organization (WHO) did not have chlorine available. PATH and its partner MSR Global Health hope the SE flow will address chlorine supply chain
hurdles by enabling facilities to produce consistently chlorinated water on the spot, at the quantity needed. The device also produces an additional
benefit: cost savings. Over five years, the SE flow is estimated to save a facility 50 percent over the cost of purchasing chlorine on the market.
PATH and MSR Global Health plan to test 50 of the devices next summary to collective evidence, refine the design, and support a broader roll out.
Validation of an innovative newborn respiratory package of equipment and training*
PATH
Respiratory infections are a leading cause of infant mortality, particularly in premature infants whose lungs are not fully formed. Without strong lungs
and appropriate oxygen intake, newborns face significant challenges in healthy development, including lifelong neurological damage or blindness. Continuous
positive airway pressure (CPAP) therapy helps mitigate these challenges; however, the tools used in high-income nations to administer this therapy
have traditionally been too costly to use in low-resource settings overseas.
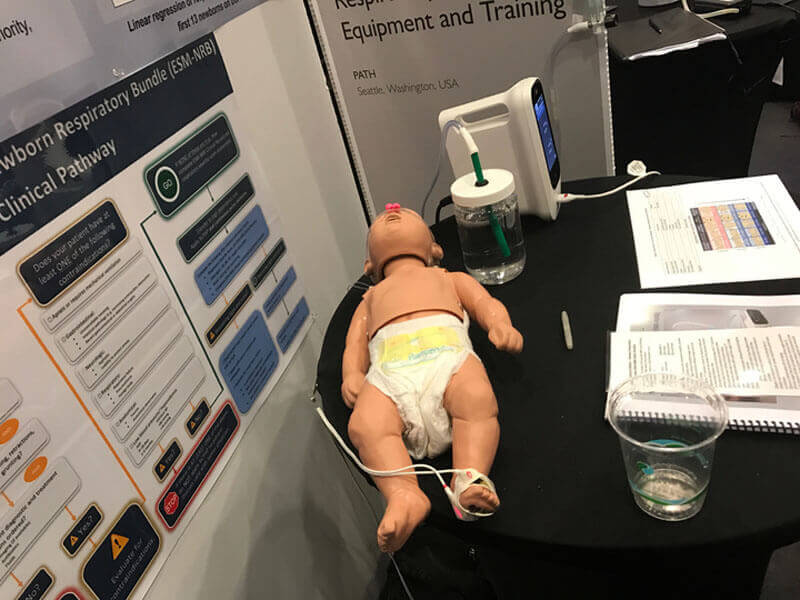 A low-cost bCPAP device for newborn health. (Photo: GHTC/Taylor Capizola) PATH and other partners developed a low-cost bubble CPAP (bCPAP) device, combined with an oxygen blender, that is appropriate for low-resource settings
and has proven effective in controlling the flow of air to support breathing. To support further uptake and effective use of this intervention, PATH
is now partnering with the Massachusetts General Hospital Division of Global Health and Human Rights to package the bCPAP, oxygen blender, nasal prongs,
and a pulse oximeter with training and educational materials, including checklists, manuals, and wall charts. The training materials will be essential
to expanding newborn access to respiratory therapy, particularly in facilities where no prior intervention existed and medical staff may be uncertain
of how to best use these new tools. The team has also partnered with Sinapi biomedical, a South African biomedical manufacturer, to manufacture the
package of tools and training materials to greatly expand the number of medical facilities using these interventions by bring the toolkit to scale.
They expect that with Saving Lives at Birth support, mortality will drop 25 percent among newborns treated in facilities introducing bCPAP therapy
for the first time.
A low-cost bCPAP device for newborn health. (Photo: GHTC/Taylor Capizola) PATH and other partners developed a low-cost bubble CPAP (bCPAP) device, combined with an oxygen blender, that is appropriate for low-resource settings
and has proven effective in controlling the flow of air to support breathing. To support further uptake and effective use of this intervention, PATH
is now partnering with the Massachusetts General Hospital Division of Global Health and Human Rights to package the bCPAP, oxygen blender, nasal prongs,
and a pulse oximeter with training and educational materials, including checklists, manuals, and wall charts. The training materials will be essential
to expanding newborn access to respiratory therapy, particularly in facilities where no prior intervention existed and medical staff may be uncertain
of how to best use these new tools. The team has also partnered with Sinapi biomedical, a South African biomedical manufacturer, to manufacture the
package of tools and training materials to greatly expand the number of medical facilities using these interventions by bring the toolkit to scale.
They expect that with Saving Lives at Birth support, mortality will drop 25 percent among newborns treated in facilities introducing bCPAP therapy
for the first time.
Building social accountability structures for respectful maternity care in Kenya: “RMC Virtual Space”
Population Council
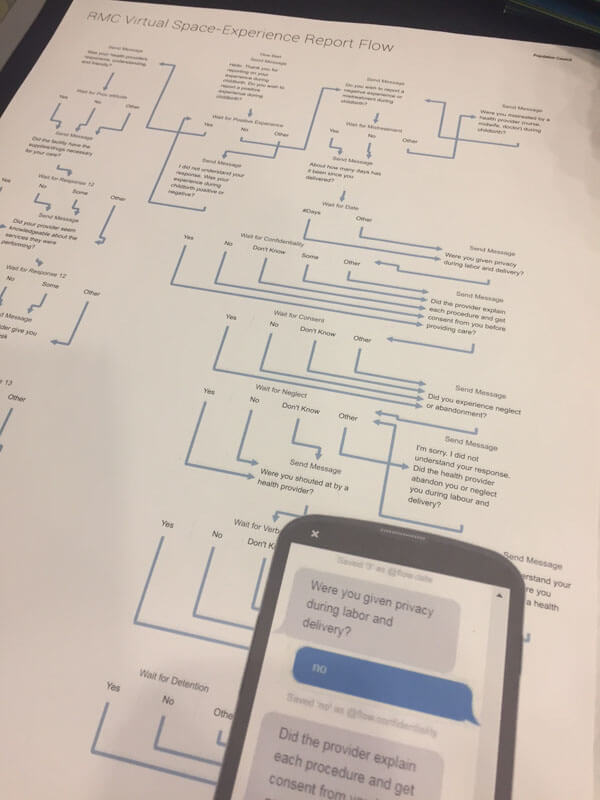 Population Council's virtual space photos. (Photo: GHTC/Marissa Chmiola)The birth of a child can be one of the most moving experiences of a mother’s life, but in many low-income countries, it can also expose a woman to abuse
and disrespect. Women who visit a health care facility during childbirth often face mistreatment, whether in the form of physical abuse, non-consent
for physical examinations, neglect, or disrespectful comments. Often women are hesitant to report these incidents due to fear of reprisal or distrust
that their feedback will be heard.
Population Council's virtual space photos. (Photo: GHTC/Marissa Chmiola)The birth of a child can be one of the most moving experiences of a mother’s life, but in many low-income countries, it can also expose a woman to abuse
and disrespect. Women who visit a health care facility during childbirth often face mistreatment, whether in the form of physical abuse, non-consent
for physical examinations, neglect, or disrespectful comments. Often women are hesitant to report these incidents due to fear of reprisal or distrust
that their feedback will be heard.
Population Council has designed a low-cost mobile text message reporting platform that they believe will empower women to report their experiences and
help providers comprehend the scope of the problem and develop solutions. This intervention works by enrolling women in the platform when they visit
a facility for care. Following a visit, the patient is prompted through text message with a series of simple questions to collect information about
her experience. The data is then aggregated and presented to the provider or facility in an easy-to-understand, dashboard-style system. Population
Council chose to design the platform using text messaging as the basis for collecting feedback because it is ubiquitous across the African continent
and provides a level of anonymity that encourages women to report their experience. The project team is at the concept stage and is currently testing
the intervention in health facilities in Kenya to solicit feedback and refine its approach.
Innovative packaging and pasteurization for donor human milk to improve health outcomes for preterm infants
PATH
Human milk banks play a critical role in ensuring that safe, high-quality human milk can be made available to infants whose mothers are unable to directly
provide their own milk. Current guidelines for human milk pasteurization require temperature control of 62.5 degrees Celsius for 30 minutes to ensure
all pathogens and bacteria are killed. Unfortunately, while this temperature kills microorganisms that can make infants sick, it also kills vital nutrients
for an infant’s growth and development. No further international guidelines have been established, leaving engineers and scientists with the challenge
of delivering safe, yet properly nutritious donor milk to milk banks.
To solve this issue, PATH is seeking to improve the two processes involved in transporting human milk to milk banks: Pasteurization and packaging. PATH
plans to research and narrow in on the optimal temperature range and timing capable of keeping human milk safe to drink and properly nutritious for
infants. As part of this, PATH is also exploring the most effective packaging that help in achieving this temperature range and timing. Currently,
the most common packaging for human milk is plastic or glass jars, but PATH researchers have found these jars do not transfer heat well or consistently.
PATH plans to test a number of packaging options from stainless steel jars, which are commonly used in countries like India, to sterile plastic bags
coated in aluminum—which in early testing have shown to distribute heat evenly and are easy to transport. Once the optimal temperature rage and
optimal packaging are discovered, PATH plans to distribute this information widely to milk banks, donor human milk pasteurization equipment manufacturers,
and human milk packagers to improve health and survival outcomes for all infants who receive heat-treated donor human milk globally.
Advancing a simple, low-cost rapid diagnostic test for urinary adipsin: A promising new biomarker-based test for improving preclampsia screening in low-resource settings*
PATH
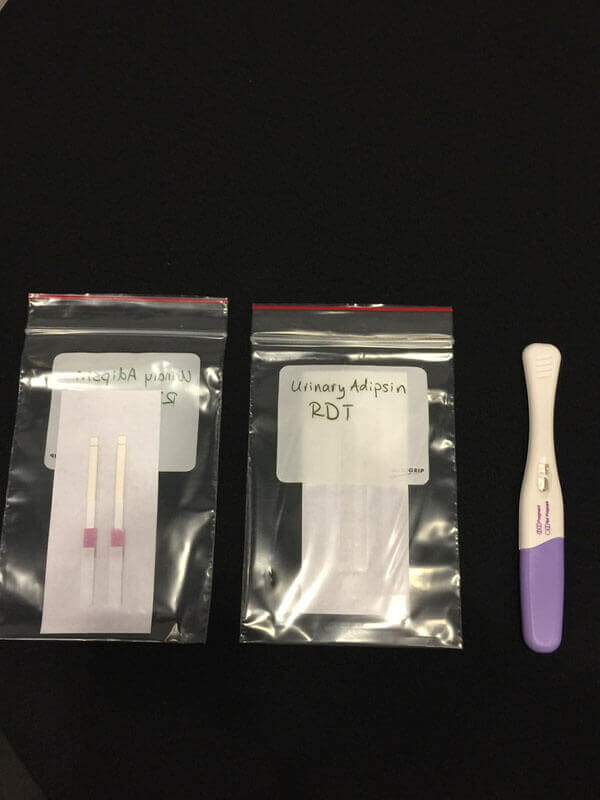 The preeclampsia (PE) test. (Photo: GHTC/Matt Robinson) Preeclampsia (PE) is a dangerous condition that causes organ failure and seizures, and it is one of the leading causes of maternal and newborn deaths worldwide.
While diagnostic tests already exist that can provide early warning of risk factors, accurate tests require samples collected over 24 hours, and rapid
tests were able to identify only 51 percent to 68 percent of risk cases. Thanks to a Saving Lives at Birth Round 4 Seed Award, PATH has developed an
early prototype of a rapid diagnostic test (RDT) that is just as fast as existing RDTs, but is capable of catching 85 percent of risk cases early in
the third trimester of pregnancy in plenty of time to take action. It is also temperature stable at 37 to 45 degrees Celsius and can be produced for
pennies on the dollar of the cost of previous tests.
The preeclampsia (PE) test. (Photo: GHTC/Matt Robinson) Preeclampsia (PE) is a dangerous condition that causes organ failure and seizures, and it is one of the leading causes of maternal and newborn deaths worldwide.
While diagnostic tests already exist that can provide early warning of risk factors, accurate tests require samples collected over 24 hours, and rapid
tests were able to identify only 51 percent to 68 percent of risk cases. Thanks to a Saving Lives at Birth Round 4 Seed Award, PATH has developed an
early prototype of a rapid diagnostic test (RDT) that is just as fast as existing RDTs, but is capable of catching 85 percent of risk cases early in
the third trimester of pregnancy in plenty of time to take action. It is also temperature stable at 37 to 45 degrees Celsius and can be produced for
pennies on the dollar of the cost of previous tests.
With its round seven award, the PATH team will undertake additional activities to bring the new RDT closer to broad scale use. First, the team will further
optimize the test to expand its shelf life and determine the feasibility of increasing the test’s accuracy. Second, the team will explore whether the
test—which currently requires a strip be dipped into urine in a separate container—can be incorporated into a form similar to at home pregnancy
tests, making it easier to use and allowing women to take the test in the comfort of their own homes. Finally, the team will begin negotiations with
commercial partners to scale up the product so it becomes widely available worldwide.
The transformative power of innovation
GHTC congratulates both our members and the other teams who were selected as finalists and award winners. This year’s prize winners will join the growing community of over 100 innovators supported by the Saving Lives at Birth partnership. Many of these innovations are beginning to have an impact as they are brought to scale. To date, the program has supported 1.5 million women and newborns and saved nearly 10,000 lives.
The Saving Lives at Birth program proves that through creative partnerships and innovative ideas and interventions, we can create dramatic improvements
in the health and well-being of people around the world.
*indicates this intervention was among the 15 winners.
GHTC staff including Taylor Capizola, program assistant; Matthew Robinson, MA, multilateral advocacy and policy officer; and Courtney Carson; US advocacy and policy officer, contributed to this article.




